SOP Reference: MWS-M03-L3
Lesson 3: Biofilm Disruption Protocols
Mechanisms for breaking down bacterial biofilms in stagnant wounds using high-MGO honey and adjunctive mechanical strategies
Clinical Context
Bacterial biofilms represent the primary obstacle to chronic wound healing. Unlike planktonic bacteria that float freely in wound fluid, biofilm-embedded organisms adhere to the wound base and secrete extracellular polymeric substances (EPS) that create a protective matrix. This architecture confers resistance to antimicrobials at concentrations up to 1000 times higher than those effective against planktonic cells.
An estimated 60 to 90 percent of chronic wounds harbor biofilms, yet conventional wound swabs detect only surface contaminants and fail to sample the adherent bacterial communities. Biofilm presence should be presumed in any wound that fails to heal despite appropriate local care, exhibits persistent malodor, or shows recurrent infection after apparent clearance.
Methylglyoxal in medical-grade Manuka honey demonstrates biofilm-penetrating capacity through multiple mechanisms: direct disruption of EPS matrix proteins, osmotic dehydration of the biofilm architecture, and sustained antimicrobial activity that prevents biofilm reformation. However, honey alone cannot eradicate established biofilms. Successful treatment requires integration of mechanical disruption, sustained chemical pressure, and repeated applications to prevent recurrence.
Biofilm Identification
Clinical Presentation Criteria
Biofilms are not visible to the naked eye at low magnification. However, characteristic clinical signs allow presumptive identification without specialized laboratory equipment. The following indicators suggest biofilm presence:
- Gelatinous or Slimy Appearance: Shiny, translucent coating on wound bed that reforms within 24 hours after mechanical removal
- Persistent Erythema: Chronic inflammation that does not respond to standard antimicrobial dressings
- Stalled Healing: Wound dimensions remain static for more than 4 weeks despite adequate debridement and infection control
- Recurrent Infection: Repeated episodes of clinical infection requiring systemic antibiotics with temporary improvement followed by relapse
- Low-Level Persistent Bioburden: Wound cultures show less than 10 to the 5th power colony-forming units per gram (subthreshold for infection) yet wound demonstrates signs of inflammation
- Silver Sulfadiazine Failure: Lack of response to topical silver preparations, which effectively suppress planktonic bacteria but penetrate biofilms poorly
Common Biofilm-Forming Organisms in Chronic Wounds
| Organism | Biofilm Characteristics | Clinical Association |
|---|---|---|
| Staphylococcus aureus | Dense EPS matrix rich in polysaccharide adhesins | Diabetic foot ulcers, surgical site infections |
| Pseudomonas aeruginosa | Alginate-based biofilm with green pigmentation | Burn wounds, pressure injuries with undermining |
| Enterococcus faecalis | Aggregative biofilm with antibiotic efflux pumps | Perianal and perineal wounds, radiation ulcers |
| Proteus mirabilis | Swarming motility biofilm with urease production | Leg ulcers in patients with urinary incontinence |
| Polymicrobial Communities | Synergistic mixed-species biofilms with enhanced resistance | Venous leg ulcers, pressure injuries in institutional settings |
Mechanical Biofilm Disruption
Physical Removal as Essential First Step
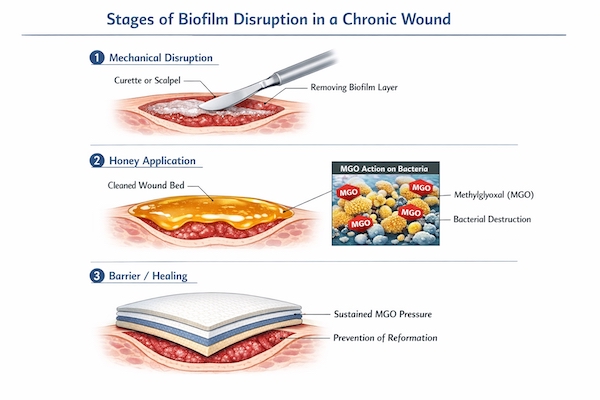
No topical antimicrobial, including high-MGO honey, can eradicate biofilm without prior mechanical disruption of the EPS matrix. The initial treatment must physically remove the majority of biofilm architecture to expose underlying bacteria to honey's antimicrobial activity. This requires aggressive but controlled debridement.
Sharp Debridement Protocol for Biofilm
In clinical settings where sharp debridement is within scope of practice, the following technique provides optimal biofilm removal:
- Anesthetize the wound bed if viable tissue must be debrided (lidocaine 1% infiltration at wound margins)
- Using sterile scalpel or curette, firmly scrape the entire wound surface to mechanically disrupt biofilm architecture
- Apply sufficient pressure to remove the gelatinous biofilm layer while avoiding deep tissue injury
- Irrigate copiously with normal saline at 8 to 15 pounds per square inch pressure (60 milliliter syringe with 18-gauge catheter)
- Repeat scraping and irrigation until wound base appears clean without visible slime reformation
- Pat dry with sterile gauze before honey application
Alternative Mechanical Methods
For settings where sharp debridement exceeds practitioner scope or patient tolerance, alternative mechanical disruption includes ultrasonic debridement (low-frequency ultrasound disrupts biofilm through cavitation), monofilament fiber pads (gentle abrasion for fragile wound beds), and pulsed lavage (mechanical irrigation at controlled pressure). All methods share the goal of physically disrupting biofilm before chemical treatment.
Methylglyoxal Anti-Biofilm Mechanisms
EPS Matrix Degradation
Methylglyoxal disrupts biofilm through multiple pathways distinct from its planktonic antibacterial activity. At concentrations above 400 milligrams per kilogram, MGO demonstrates the following anti-biofilm effects:
- Protein Glycation: MGO reacts with lysine and arginine residues in EPS matrix proteins, causing structural destabilization and matrix collapse
- Quorum Sensing Disruption: MGO interferes with bacterial communication molecules (autoinducers) that coordinate biofilm development and antibiotic resistance gene expression
- Osmotic Stress: High sugar content creates hyperosmolar environment that dehydrates biofilm matrix and increases antibiotic penetration
- Reactive Oxygen Species Generation: MGO metabolism by bacteria produces hydrogen peroxide, creating oxidative stress that damages biofilm-embedded organisms
- eDNA Degradation: Extracellular DNA serves as structural scaffold in some biofilms; honey's peroxidases degrade eDNA and weaken biofilm integrity
Concentration Requirements for Biofilm Activity
Biofilm eradication requires sustained exposure to MGO concentrations substantially higher than those needed for planktonic bacterial suppression. Standard honey therapy at 100 to 250 milligrams per kilogram prevents biofilm formation in clean wounds but cannot disrupt established biofilms. Biofilm treatment protocols mandate 400 to 1000 milligrams per kilogram MGO applied continuously for 14 to 21 days.
Anti-Biofilm Treatment Protocol
Phase I: Aggressive Disruption (Days 1 to 7)
The initial week focuses on maximal biofilm removal and prevention of immediate reformation. This phase combines mechanical and chemical strategies at their most intensive application.
- Day 1: Perform sharp or ultrasonic debridement to remove visible biofilm. Apply 600 to 1000 mg/kg MGO honey in thick layer (5 millimeters). Cover with non-adherent contact layer and absorbent secondary dressing.
- Day 2: Remove dressing. Irrigate wound with normal saline. Apply fresh honey at same concentration without repeat debridement unless biofilm visibly reformed.
- Day 3: Remove dressing. If gelatinous coating has returned, repeat mechanical disruption with monofilament pad. Reapply high-concentration honey.
- Days 4-7: Continue daily honey applications. Perform mechanical disruption every 48 hours if biofilm reformation visible. Goal is to prevent biofilm re-establishment through sustained chemical pressure.
Phase II: Suppression and Transition (Days 8 to 21)
Once biofilm no longer visibly reforms within 24 hours, transition to suppression phase focused on preventing recurrence while supporting granulation.
Reduce dressing change frequency to every 48 hours. Continue high-concentration honey (400 to 600 mg/kg) but decrease layer thickness to 3 millimeters. Monitor for biofilm recurrence by assessing wound bed appearance before each dressing change. Any return of gelatinous coating requires immediate escalation back to Phase I protocol.
Phase III: Maintenance (Day 22 Onward)
After three weeks without biofilm reformation, de-escalate to standard wound healing protocols. Reduce MGO concentration to 250 to 400 mg/kg. Extend dressing changes to every 72 hours. Transition focus from biofilm suppression to epithelialization support.
Adjunctive Biofilm Control Strategies
Negative Pressure Wound Therapy Integration
Negative pressure wound therapy (NPWT) enhances biofilm control through mechanical stress on bacterial communities and continuous removal of planktonic bacteria released during biofilm disruption. When combined with honey therapy, apply honey to wound bed, place non-adherent interface layer, then apply NPWT foam and initiate suction at 75 to 125 millimeters mercury continuous pressure. Change NPWT dressing every 48 to 72 hours with fresh honey application at each change.
Antimicrobial Stewardship Considerations
Systemic antibiotics do not penetrate biofilms effectively and should not be used as primary biofilm treatment. Reserve systemic therapy for documented invasive infection (cellulitis, osteomyelitis, bacteremia) rather than biofilm suppression. Topical antiseptics such as cadexomer iodine or polyhexamethylene biguanide may be used in combination with honey during Phase I if biofilm proves particularly resistant, but avoid long-term use to prevent emergence of resistance.
Patient Education on Biofilm Chronicity
Set realistic expectations with patients regarding biofilm treatment duration. Unlike acute bacterial infections that resolve in days, biofilm eradication requires weeks of consistent therapy. Explain that visible wound improvement may lag behind microbiological clearance by 7 to 14 days. Premature discontinuation of high-concentration honey before biofilm eradication allows rapid reformation and treatment failure.
Treatment Failure Recognition
Indicators of Inadequate Biofilm Control
Despite appropriate protocol adherence, some biofilms resist honey-based disruption. The following signs suggest treatment modification or escalation to surgical intervention:
- Persistent Biofilm Reformation: Visible return of gelatinous coating within 12 hours despite mechanical disruption and high-MGO honey
- Increasing Wound Dimensions: Progressive enlargement despite biofilm treatment suggests deeper infection or tissue ischemia
- Bone Exposure Development: New areas of exposed bone indicate possible osteomyelitis requiring surgical debridement
- Constitutional Symptoms: Fever, elevated white blood cell count, or sepsis physiology indicates systemic infection requiring hospital admission
- Fistula Formation: Development of sinus tracts or undermining suggests abscess or foreign body requiring surgical exploration
Escalation Pathways
When honey-based biofilm control fails, consider the following interventions: surgical debridement to bone (if osteomyelitis suspected), tissue biopsy for quantitative culture and histopathology, advanced imaging (MRI or bone scan) to identify occult abscess or bone involvement, infectious disease consultation for biofilm-active antibiotics (such as rifampin or fosfomycin), or wound closure procedures (flap, graft, amputation) if limb salvage not achievable with local care.
Critical Takeaways
- Biofilms are present in 60 to 90% of chronic wounds and confer up to 1000-fold antibiotic resistance
- Mechanical disruption via sharp debridement or ultrasonic methods is essential before chemical treatment
- High MGO concentrations (600 to 1000 mg/kg) required for biofilm eradication, not just suppression
- Treatment requires 14 to 21 days of sustained high-concentration honey with frequent mechanical re-disruption
- Daily dressing changes during Phase I (days 1 to 7) prevent biofilm reformation during vulnerable period
- Persistent biofilm despite protocol adherence indicates need for surgical intervention or advanced imaging