SOP Reference: MWS-M03-L1
Lesson 1: Autolytic Debridement Protocols
Selective removal of necrotic tissue using high-MGO honey substrates while preserving viable wound bed architecture
Clinical Context
Autolytic debridement leverages the body's endogenous proteolytic enzymes to selectively digest devitalized tissue. Medical-grade Manuka honey creates a moisture-rich wound environment that supports autolysis while simultaneously delivering antimicrobial activity via methylglyoxal. This dual-action mechanism distinguishes honey-mediated debridement from purely passive moist wound healing approaches.
Traditional sharp debridement removes nonviable tissue through mechanical instrumentation, requiring specialized training and carrying risks of iatrogenic damage to healing tissue. Enzymatic debridement using collagenase preparations targets specific protein substrates but may digest viable extracellular matrix components. Autolytic debridement with high-MGO honey offers a middle path: selective, patient-tolerable, and compatible with outpatient wound care protocols.
The protocol described here applies specifically to chronic wounds exhibiting slough, eschar, or devitalized tissue that impedes epithelialization. Acute traumatic wounds, surgical incisions healing by primary intention, and burns requiring specialized escharotomy fall outside this clinical scope.
Wound Assessment Prerequisites
Baseline Evaluation Before Initiating Debridement
Autolytic debridement protocols require baseline wound characterization to establish appropriate treatment parameters and define endpoints. The following assessments occur during the initial evaluation:
- Tissue Viability Mapping: Document the percentage of necrotic versus granulating tissue using standardized photographic wound imaging with metric scale reference
- Wound Dimensions: Record length, width, and depth in millimeters at the widest points, noting any undermining or tunneling
- Exudate Volume Classification: Assign a qualitative descriptor (none, minimal, moderate, copious) based on dressing saturation between changes
- Perfusion Status: Assess capillary refill in periwound tissue and document presence or absence of palpable pulses in affected extremity
- Infection Indicators: Record presence of purulence, perilesional erythema, warmth, edema, or odor that may indicate biofilm or invasive infection
- Pain Assessment: Establish baseline pain scores using numeric rating scale (0 to 10) at rest and during dressing changes
Contraindications to Honey-Mediated Debridement
The following clinical scenarios preclude honey application without additional medical consultation or intervention:
| Contraindication | Clinical Concern | Required Intervention |
|---|---|---|
| Dry Gangrene | Eschar maintains vascular barrier preventing systemic infection spread | Vascular surgery consult for revascularization before debridement |
| Exposed Bone/Tendon | Osmotic drawing effect may desiccate exposed structures | Surgical debridement with flap coverage required first |
| Active Bleeding | Moisture environment impairs hemostasis | Achieve hemostasis with pressure or cautery before honey application |
| Known Honey Allergy | Risk of contact dermatitis or systemic hypersensitivity | Alternative autolytic agent (hydrogel) or enzymatic debridement |
| Critical Limb Ischemia | Inadequate perfusion prevents healing despite debridement | Revascularization prior to wound optimization |
MGO Concentration Selection
Matching Honey Potency to Wound Burden
Medical-grade Manuka honey ranges from 100 to 1000 milligrams methylglyoxal per kilogram. Higher concentrations deliver more aggressive antimicrobial activity but generate greater osmotic draw, which can increase exudate production and patient discomfort. The optimal MGO concentration balances debridement efficacy against tolerability.
| MGO Range (mg/kg) | Clinical Indication | Autolytic Timeline |
|---|---|---|
| 100 to 250 | Minimal slough, clean wound bed with mild bioburden | 5 to 7 days for complete autolysis |
| 250 to 400 | Moderate slough covering less than 50% of wound surface | 7 to 10 days for complete autolysis |
| 400 to 600 | Heavy slough or thin eschar, suspected biofilm presence | 10 to 14 days for complete autolysis |
| 600 to 1000 | Thick eschar, necrotic tissue adherent to wound base | 14 to 21 days for complete autolysis |
Concentration Adjustment Protocol
If debridement progress stalls after one week of treatment with no visible reduction in necrotic tissue burden, step up to the next MGO tier. Conversely, if the patient reports intolerable pain or excessive exudate requiring daily dressing changes, step down to a lower concentration. Document all concentration adjustments in the wound care record with clinical justification.
Application Protocol
Sterile Technique Requirements
Debridement using medical-grade honey does not require surgical asepsis but mandates clean technique to prevent introduction of exogenous pathogens. The following protocol applies to outpatient wound clinic settings:
- Perform hand hygiene with antimicrobial soap or alcohol-based hand rub per CDC guidelines
- Don clean disposable gloves (sterile gloves not required for chronic wound management)
- Remove existing dressing and discard in appropriate waste receptacle
- Irrigate wound with normal saline or sterile water at body temperature (avoid cold irrigation which impairs enzymatic activity)
- Pat periwound skin dry using sterile gauze to prevent maceration from honey migration
- Apply honey directly to wound bed in a layer approximately 3 to 5 millimeters thick using a sterile tongue depressor or single-use applicator
- Ensure complete coverage of all necrotic tissue without extending beyond wound margins onto intact skin
- Cover with non-adherent primary dressing (such as petroleum-impregnated gauze or silicone contact layer)
- Apply absorbent secondary dressing sized to accommodate anticipated exudate volume
- Secure with conformable retention tape or tubular bandage, avoiding circumferential wrapping that impairs circulation
Honey Volume Calculation
The required honey volume depends on wound surface area and desired application thickness. For clinical planning, assume 1 milliliter of honey covers approximately 5 square centimeters at the standard 3-millimeter depth. A 10 square centimeter wound therefore requires approximately 2 milliliters (roughly 3 grams) of honey per application.
Monitoring and Endpoint Criteria
Expected Progression Timeline
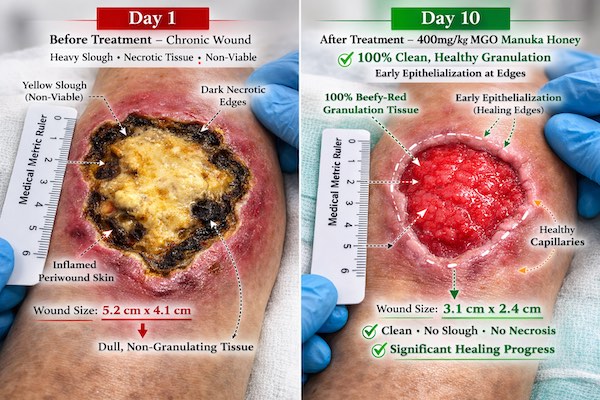
Autolytic debridement generates visible changes in wound appearance within 48 to 72 hours of initial application. Necrotic tissue softens, changes color from black or brown to yellow or gray, and begins to separate from underlying viable tissue. Complete debridement occurs when 100% of the wound bed consists of granulation tissue with no residual slough or eschar.
Clinical Indicators of Treatment Failure
If the following signs appear during debridement treatment, reassess the wound care plan and consider alternative interventions:
- Expanding Necrosis: Increase in necrotic tissue area despite honey application suggests inadequate perfusion or progressive infection requiring systemic intervention
- Foul Odor Intensification: Worsening malodor despite antimicrobial honey contact indicates anaerobic infection or retained foreign material requiring sharp debridement
- Perilesional Cellulitis: Expanding erythema, warmth, or induration beyond wound margins indicates systemic antibiotic requirement
- Undermining Extension: Progressive undermining or sinus tract development suggests osteomyelitis or retained abscess requiring imaging and surgical consultation
- Patient Intolerance: Persistent pain exceeding baseline levels by more than 3 points on numeric scale warrants concentration reduction or modality change
Documentation Requirements
Each dressing change requires documentation of wound dimensions, tissue type percentages, exudate volume, periwound skin integrity, patient pain score, and photographic evidence using standardized wound imaging protocol. These serial assessments establish treatment efficacy and provide medicolegal protection in the event of adverse outcomes.
Transition to Granulation Phase
Recognizing Debridement Completion
The endpoint of autolytic debridement occurs when necrotic tissue has been completely removed and the wound bed displays healthy granulation tissue characterized by beefy red coloration, moist surface, and absence of slough. At this transition point, treatment objectives shift from debridement to epithelialization support.
Continue honey application during the granulation phase, but reduce MGO concentration to the 100 to 250 milligram per kilogram range. Higher concentrations become unnecessary once bioburden is controlled and may impair keratinocyte migration across the wound surface. Dressing change frequency can decrease from daily to every 48 to 72 hours as exudate production diminishes.
Critical Takeaways
- Autolytic debridement with Manuka honey selectively removes necrotic tissue while preserving viable wound bed architecture
- MGO concentration ranges from 100 to 1000 mg/kg depending on wound burden, with higher concentrations for heavy slough or eschar
- Apply honey in 3 to 5 millimeter layer directly to necrotic tissue, avoiding intact periwound skin
- Expected debridement timeline ranges from 5 to 21 days depending on tissue type and MGO potency
- Monitor for treatment failure indicators including expanding necrosis, worsening odor, or perilesional cellulitis
- Transition to lower MGO concentrations once wound bed reaches complete granulation