SOP Reference: MWS-M03-L2
Lesson 2: Exudate Management Protocols
Osmotic pressure regulation for highly effusing chronic wounds using strategic dressing layering and honey concentration control
Clinical Context
Chronic wound exudate contains proteolytic enzymes, inflammatory cytokines, and bacterial endotoxins that impair healing when allowed to pool in the wound bed or macerate periwound skin. Medical-grade Manuka honey creates an osmotic gradient that draws fluid from the wound base into the dressing matrix, but this same mechanism can generate excessive exudate in highly inflamed wounds.
The challenge in exudate management lies in maintaining optimal moisture balance. Too little moisture causes desiccation and cell death at the wound surface. Too much moisture overwhelms dressing absorptive capacity, leading to leakage, maceration, and increased dressing change frequency. Effective protocols calibrate honey concentration and dressing selection to match wound output.
Exudate volume correlates with wound bioburden, inflammation status, and venous insufficiency severity. As honey therapy reduces bacterial load and inflammatory mediators, exudate production typically decreases over the first two weeks of treatment. This dynamic requires ongoing assessment and protocol adjustment rather than static dressing regimens.
Exudate Volume Classification
Standardized Assessment Criteria
Accurate exudate quantification guides dressing selection and honey concentration decisions. The following classification system provides reproducible assessment criteria based on dressing saturation patterns:
| Classification | Clinical Appearance | Dressing Saturation |
|---|---|---|
| None | Wound bed appears dry with possible desiccation | Dressing remains completely dry at removal |
| Minimal | Moist wound bed without pooling fluid | Primary dressing shows less than 25% saturation |
| Moderate | Visible moisture with occasional small fluid accumulation | Primary dressing 25 to 75% saturated, secondary dressing dry |
| Heavy | Pooling fluid visible on wound surface | Primary dressing 75 to 100% saturated, secondary shows partial saturation |
| Copious | Continuous drainage with periwound maceration | Both primary and secondary dressings fully saturated, leakage onto outer bandage |
Exudate Characteristics Documentation
Beyond volume, exudate composition provides diagnostic information. Document color (serous, serosanguinous, sanguinous, purulent), viscosity (thin, thick, gelatinous), and odor (none, mild, moderate, foul) at each dressing change. Purulent exudate with foul odor indicates infection requiring culture and possible systemic antibiotics. Serous exudate progression from copious to moderate volume signals treatment efficacy.
Osmotic Balance Strategy
Honey Concentration and Exudate Production
Medical-grade honey functions as a hyperosmolar agent, creating an osmotic gradient of approximately 600 to 1200 milliosmoles per kilogram depending on MGO concentration and water activity. This gradient drives fluid from the wound bed into the dressing, effectively debriding the wound but simultaneously increasing short-term exudate output.
In highly exudating wounds, the paradox becomes apparent: the honey that provides antimicrobial benefit also generates more fluid to manage. The solution involves staged concentration adjustment and strategic dressing architecture.
Initial Phase Protocol (Days 1 to 7)
For wounds classified as heavy or copious exudators, begin with lower MGO concentrations (100 to 250 milligrams per kilogram) despite potential bioburden. This minimizes osmotic draw while still delivering antimicrobial activity. Accept that debridement will progress more slowly in exchange for manageable exudate volumes.
Apply honey in a thinner layer (approximately 2 millimeters) compared to standard debridement protocol. This reduces the total osmotic load while maintaining wound bed coverage. Pair with highly absorbent secondary dressings capable of managing the anticipated fluid volume.
Escalation Phase Protocol (Days 7 to 14)
As exudate volume decreases in response to bioburden reduction, incrementally increase MGO concentration to enhance debridement efficacy. Move from 100 to 250 milligrams per kilogram up to 250 to 400 milligrams per kilogram once dressing saturation patterns indicate the wound can tolerate the increased osmotic gradient.
The goal is to reach therapeutic MGO levels for optimal antimicrobial activity without overwhelming dressing absorptive capacity. This titration approach prevents the treatment-induced maceration that occurs when high-concentration honey is applied to highly exudating wounds from the outset.
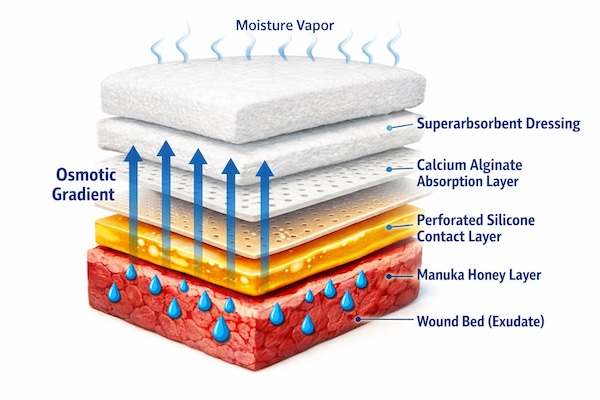
Dressing Architecture for High-Output Wounds
Multi-Layer Absorption System
Managing copious exudate requires a strategic layering approach that moves fluid away from the wound bed through progressive absorption zones. The following architecture provides optimal fluid handling:
- Primary Contact Layer: Medical-grade honey applied at 2 to 3 millimeter depth directly to wound bed
- Interface Layer: Non-adherent perforated film (such as silicone contact layer) prevents honey migration onto periwound skin while allowing vertical fluid transmission
- Absorption Layer 1: Calcium alginate or hydrofiber dressing directly over interface layer to handle the primary exudate load and form gel matrix that locks fluid
- Absorption Layer 2: Superabsorbent polymer dressing or thick foam capable of handling overflow from Layer 1
- Outer Retention Layer: Conformable stretch gauze or tubular bandage to secure all layers without compression
Dressing Change Frequency Calibration
The absorption system should maintain integrity for the target wear time without saturation breakthrough. For heavy exudators, this typically means daily changes during the first week. As exudate decreases, extend to every 48 hours, then 72 hours as wound transitions to moderate output.
If dressings saturate before the planned change interval, reassess the absorption architecture rather than simply increasing change frequency. Adding an additional superabsorbent layer often proves more effective than doubling dressing changes.
Periwound Skin Protection
Maceration Prevention Strategies
Chronic exposure to wound exudate causes maceration, characterized by white, wrinkled, fragile skin at wound margins. Macerated skin breaks down easily, enlarging the wound and increasing infection risk. Prevention requires barrier application before honey dressing placement.
- Liquid Barrier Films: Apply cyanoacrylate-based or polymer-based skin protectant to intact periwound skin, creating a moisture-impermeable barrier that prevents exudate contact
- Hydrocolloid Strips: Cut thin hydrocolloid dressing into strips and frame the wound edges, creating a raised border that blocks lateral fluid migration
- Zinc Oxide Paste: Traditional but effective, zinc oxide creates a physical barrier with mild antimicrobial properties; best for wounds with very heavy exudate
- Petrolatum-Impregnated Gauze: Placed only on periwound skin (not in wound bed) to repel moisture away from fragile epithelium
Treatment of Established Maceration
If maceration develops despite preventive measures, immediately reduce honey concentration and improve absorption capacity. Apply zinc oxide paste to macerated areas and avoid any adhesive products that could traumatize fragile skin. In severe cases, temporarily discontinue honey and allow periwound skin to recover with dry gauze dressings before resuming treatment at lower concentration.
Venous Insufficiency Considerations
Compression Therapy Integration
Venous leg ulcers produce copious exudate driven by elevated capillary hydrostatic pressure. Honey dressings address the wound surface environment, but without compression therapy to correct the underlying venous hypertension, exudate will remain excessive regardless of dressing selection.
Apply medical-grade honey and primary dressings first, then apply therapeutic compression (30 to 40 millimeters mercury at the ankle) over the dressing system. Compression reduces capillary leakage, decreases edema-driven exudate production, and accelerates healing through improved venous return.
Compression Bandage Selection
Multi-layer compression systems work synergistically with honey dressings. The inner padding layer absorbs excess exudate that overwhelms the primary dressing. The outer compression layers provide sustained graduated pressure. Change the honey dressing per protocol (daily to every 3 days) while leaving compression layers intact for one week unless saturated.
| Compression Type | Application | Wear Duration |
|---|---|---|
| Four-Layer Bandage System | High exudate venous ulcers with edema | 7 days (change honey dressing through window or remove/reapply) |
| Short-Stretch Bandage | Moderate exudate with normal ankle mobility | 7 days with daily honey dressing access |
| Compression Hosiery (20 to 30 mmHg) | Minimal exudate maintenance phase | Daily removal for honey application |
| Tubular Compression | Light support for low exudate wounds | Change with each honey dressing |
Monitoring and Adjustment Criteria
Expected Exudate Trajectory
Successful honey therapy produces a predictable exudate reduction pattern. Heavy exudators should decrease to moderate output within 7 to 10 days. Moderate exudators should reach minimal output by 14 days. Wounds that maintain heavy or copious exudate beyond 14 days despite appropriate honey concentration and dressing selection require reassessment for untreated infection, occult abscess, or inadequate compression.
Red Flag Indicators
The following scenarios indicate treatment failure or complication requiring intervention:
- Increasing Exudate Volume: Progressive increase in output despite honey therapy suggests worsening infection or inflammatory response
- Purulent Character Development: Transition from serous to purulent exudate indicates bacterial overgrowth requiring culture and possible systemic antibiotics
- Foul Odor Persistence: Continued malodor beyond 48 hours of honey treatment suggests anaerobic infection or biofilm that honey concentration cannot overcome
- Periwound Cellulitis: Expanding erythema, warmth, or edema indicates infection spread requiring systemic treatment
- Patient Constitutional Symptoms: Fever, chills, or malaise suggests systemic infection requiring urgent medical evaluation
Critical Takeaways
- Medical-grade honey creates osmotic gradient that increases short-term exudate production while reducing long-term output through bioburden control
- Start highly exudating wounds with lower MGO concentrations (100 to 250 mg/kg) to minimize osmotic draw
- Multi-layer absorption systems handle heavy exudate through calcium alginate, superabsorbent polymers, and foam dressings
- Protect periwound skin with barrier films or zinc oxide paste to prevent maceration
- Integrate compression therapy for venous ulcers to address underlying cause of excessive exudate
- Expect exudate reduction from heavy to moderate within 7 to 10 days; persistent heavy output indicates treatment failure